vph
News, Guidelines, Etc..

FSMA Food Safety Modernization Act
Building a new food safety system based on prevention will take time, and FDA is creating a process for getting this work done. Congress has established specific implementation dates in the legislation. The funding the Agency gets each year, which affects staffing and vital operations, will affect how quickly FDA can put this legislation into effect. FDA is committed to implementing the requirements through an open process with opportunity for input from all stakeholders.
For the first time, FDA will have a legislative mandate to require comprehensive, science-based preventive controls across the food supply. This mandate includes:
- Mandatory preventive controls for food facilities: Food facilities are required to implement a written preventive controls plan. This involves: (1) evaluating the hazards that could affect food safety, (2) specifying what preventive steps, or controls, will be put in place to significantly minimize or prevent the hazards, (3) specifying how the facility will monitor these controls to ensure they are working, (4) maintaining routine records of the monitoring, and (5) specifying what actions the facility will take to correct problems that arise. (Final rule due 18 months following enactment)
-
Mandatory produce safety standards: FDA must establish science-based, minimum standards for the safe production and harvesting of fruits and vegetables. Those standards must consider naturally occurring hazards, as well as those that may be introduced either unintentionally or intentionally, and must address soil amendments (materials added to the soil such as compost), hygiene, packaging, temperature controls, animals in the growing area and water. (Final regulation due about 2 years following enactment)
Authority to prevent intentional contamination: FDA must issue regulations to protect against the intentional adulteration of food, including the establishment of science-based mitigation strategies to prepare and protect the food supply chain at specific vulnerable points. (Final rule due 18 months following enactment).
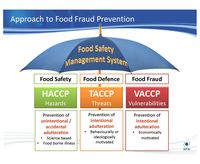
TACCP, What is it?
TACCP is a method, partly similar in tools and techniques to those used with HACCP
(Hazard Analysis and Critical Control Points), that assesses hazards and risks to the business, process or product from attack for malicious purposes, fraud, or gain for individuals or groups at the expense of the targeted organisation. The threat or attack may come from either internal or external sources.
Some of the aspects of TACCP that are different to HACCP form an integral part in the additional validation work that will be necessary to prove that the systems and protocols put in place are capable of defending the organisation.
The potential hazards may detail not only chemical, physical and biological hazards as with a conventional HACCP study, but also might cover the elements of radiological hazards and of adulteration and/or sabotage. Sabotage particularly is difficult to assess and evaluate, therefore making it harder to control and validate. What can be validated, however, are the control mechanisms in place, and the fact that all potential points of attack are dealt with in the study.